Successful organ transplantation hinges on finding a match, both in size and biology, and one of the most important factors is a compatible blood type. A new approach shows promise in converting whole type A lungs into the ‘universal’ type O.
This innovative protocol could mean more fair distribution of donor organs to the sickest patients.
The study was led by researchers at Toronto’s University Health Network (UHN) and included contributions from the University of Toronto, University of British Columbia, and University of Alberta. The work was published in Science Translational Medicine.
“With the current matching system, wait times can be considerably longer for patients who need a transplant depending on their blood type,” said senior author Marcelo Cypel, thoracic surgeon at UHN and professor of surgery at the University of Toronto, in a press release.
“Having universal organs means we could eliminate the blood-matching barrier and prioritize patients by medical urgency, saving more lives and wasting less organs.”
Why blood type matters in organ transplantation
A person’s blood type depends on whether they display certain sugars on their red blood cells. These sugars can trigger an immune response, so they are called antigens.
There are two main blood antigens — antigen A and antigen B — that lead to four main blood groups:
- type A with antigen A
- type B with antigen B
- type AB with both antigens A and B
- and type O that carries neither
The body will only make antibodies against the blood antigens it doesn’t have. Those antibodies serve to detect foreign cells and mark them for destruction, while leaving its own cells alone — similar to how antibodies against bacteria or viruses help the body recognize and respond to an infection.
Those same antigens line the blood vessels of all our organs, as well. So putting type A lungs into a type O patient would trigger a swift and potentially life-threatening immune attack. But type O lungs have neither antigen and are the most likely to be compatible with any patient, as long as the size also matches.
Converting lungs to the universal donor type
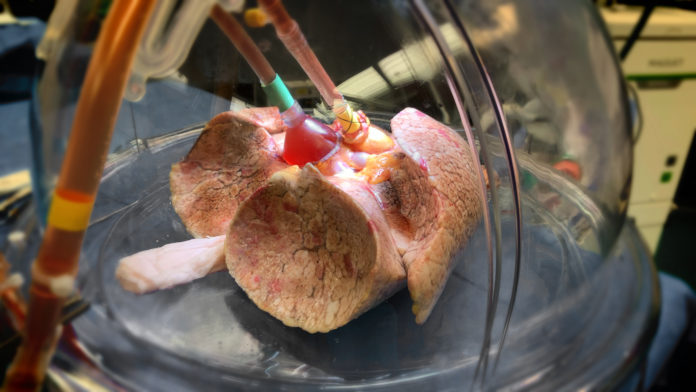
The innovation features various aspects of past research from across Canada. Cypel’s research team at UHN developed ex vivo lung perfusion (EVLP), a method that has improved the state of donor lungs for over 550 patients who received transplants at UHN since 2012. They remain one of only a few centres in the world with this technology.
Donor lungs don’t tend to fare well on ice, limiting the window for successful transplantation. By contrast, EVLP places donor lungs inside a sterile protective dome where they can be ventilated and supplied with warm, oxygenated fluids and nutrients. This more life-like environment helps preserve them for longer.
Because EVLP pumps fluids through existing blood vessels, donor lungs can also be treated by using that network to distribute compounds to every pocket of tissue. In the past this has been used to treat infection or inflammation, further widening the pool of lungs suitable for transplant.
That same approach could also be used to distribute other compounds, like the gut bacteria-derived enzymes used by co-author Stephen Withers, professor of chemistry at the University of British Columbia.
Withers previously demonstrated that these enzymes — used by gut bacteria to snip sugars off the things we eat to use as food — could be isolated and used on type A blood, efficiently converting it to type O blood as measured by every standard used by Canadian Blood Services to determine blood type.
It’s relatively easy to disperse the enzymes into a container filled with blood, but pumping them throughout an entire organ had never been done before. The collaboration presented an opportunity to deliver the enzymes with EVLP to attempt just that.
The team sourced type A human lungs that were not suitable for transplant for their experiment. They found that they could remove over 97 percent of antigen A within four hours without causing acute lung toxicity.
As a test of how well the treated lungs might respond in a type O patient, whose plasma would carry both anti-A and anti-B antibodies, they then used the EVLP circuit to send type O plasma through. They looked for signs of hyperacute rejection — where the antibodies would bind to the lungs and signal an immune attack — and found that the treated lungs were likely to be tolerated.
The combination of these technologies could provide much needed equity in matching organ donors and recipients. Currently, type O patients wait on average twice as long for a lung transplant as type A patients, putting them at greater risk of dying while they wait. The team hopes to start a clinical trial within the next 12 to 18 months.