Proteins are the molecular machinery behind all living things. From transport, to digestion, to replication, to communication, proteins carry out critical tasks that keep cells running. It makes sense that they are important in health and disease.
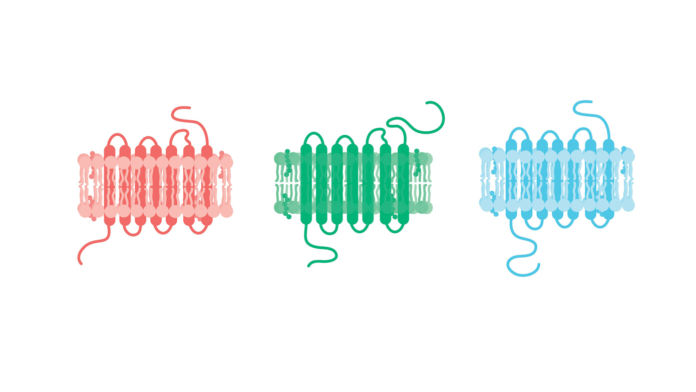
In particular, proteins that are found in the cell membrane, a fatty envelope that encloses the cell, are important in how cells communicate with one another. Defects or unintended changes in the amounts of each protein can change the way they behave.
You may have heard of the concept of a lock-and-key fit between molecules that work together. Shape is an important feature of proteins, but proteins aren’t rigid: they are constantly moving but are usually found in a handful of key signature shapes called conformations. Understanding protein shape in disease can help researchers understand how malfunctioning cells might respond to medicine.
To study the 3D shapes of important proteins, researchers have to isolate them in the lab, and until now detergents have been used to break up the cell. The problem is that the fat molecules in the cell membrane stabilize the shapes of the proteins they surround, and detergents strip these away.
Oliver Ernst, professor of biochemistry at the University of Toronto, has come up with a way to extract membrane-bound proteins without taking them out of their fatty environment. Ernst used a plastic originally developed for the auto industry called styrene–maleic acid (SMA) to break up the membrane into discs, keeping proteins together with a raft of surrounding membrane. These discs could then be used to study native 3D protein conformation using xray crystallography.
This technique opens opportunities to better understand diseases at a molecular level, and design more effective and targeted therapies to treat them.