Ebola, the deadly viral hemorrhagic fever that reached epidemic proportions in West Africa in 2014, has been making headlines again, but this time, the news is good. The Ebola vaccine being tested on at-risk individuals in Guinea was 100% effective – that is, from day 10 after vaccination and onward, not one person developed the disease. The results of the trial were published in The Lancet last week.
But this success story actually started more than 10 years ago inside the unassuming white walls of the National Microbiology Laboratory (NML) in Winnipeg.
Finding a cure before it’s needed
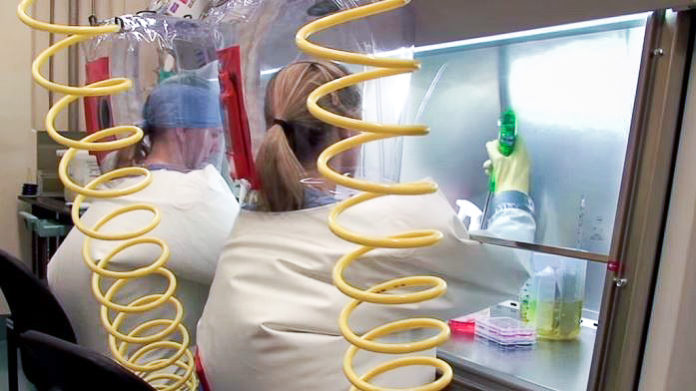
With its Level 4 biosafety rating, the NML is the only facility in Canada that can work with pathogens like Ebola – those that are potentially fatal and have no available vaccines or treatments. The Ebola virus has an average fatality rate of 50%, meaning that six to 16 days after the onset of fever, fatigue, vomiting, and in some cases, internal and external bleeding, about half of infected individuals will die.
So every day before working with the virus, scientists cover up from head to toe in protective suits that are inflated with air, making sure any airborne viral particles are blown away from the body. Before leaving the lab, scientists must decontaminate in a chemical shower.
“It’s time consuming, but also nice and quiet,” says Dr. Gary Korbinger, former Head of the Special Pathogens program at the Public Health Agency of Canada, and instrumental in the vaccine’s development.
And in that quiet, the vaccine was born.
Training the immune system
Viruses cause disease by entering a cell and hijacking the cell’s own machinery to produce millions of virus copies. Once created, the new viruses burst from the cell, killing it and quickly spreading. Like any viral infection, it’s the immune system’s job to recognize and neutralize the viral threat. But the Ebola virus contains proteins that interfere with the body’s innate immune response, allowing the virus to go unchecked. Ironically, immune cells that normally protect the body actually help shuttle the virus to the lymph nodes where it can enter the bloodstream and be fast-tracked throughout the body.
The role of the Ebola vaccine is to train the immune system to recognize the Ebola virus before it can wreak havoc on our bodies. In this case, the vaccine contains an animal virus, not associated with disease in humans, to jump start the immune system, and one protein from the Ebola virus’ protective coating to act as a future guide.
It’s the choice of viral carrier and Ebola protein that is key – not all combinations are equally effective.
Over 15 years, the team at NML tested more than 50 different vaccine strategies with at least 10 virus platforms.
Then finally, in 2005, they found the right one. With just on injection, this particular combination, called rVSV-ZEBOV, completely protected non-human primates from contracting Ebola after exposure to the virus.
The road to trial
The Canadian government patented the vaccine and produced around 1,000 vials, but it wasn’t until 2010 that it was licensed to NewLink Genetics, a US-based biopharmaceutical company. There, development stalled, perhaps because Ebola is not as widespread as many other diseases, with relatively rare outbreaks only affecting a few hundred people each time.
But the devastation of the 2014 outbreak provided all the incentive and funding needed to produce clinically-usable rVSV-ZEBOV. NewLink entered into an exclusive worldwide licensing agreement Merck & Co to get the vaccine up and running. Soon after, rVSV-ZEBOV was chosen by the World Health Organization for this trial, as the most promising Ebola vaccine candidate in the pipeline.
But the success of rVSV-ZEBOV is farther reaching than the results of this one trial.
“The same approach is easily adaptable to Sudan, Marburg, Lassa, and other dangerous viruses,” says Korbinger. He likens this vaccine to a cookbook that provides a successful base vaccine recipe.
Korbinger is now at Université Laval working on more vaccines, this time for viruses such as HIV that cause chronic infections.
Perhaps Canadians will be at the forefront of the next vaccine success story as well.